肠道是动物吸收营养物质的主要器官,也是保障机体免受病原菌和毒素侵害的生理屏障[1]。处于幼龄阶段的动物,由于其消化道发育不全,被动免疫力不足,更容易遭受有害菌攻击,产生肠道疾病,导致营养物质吸收利用率下降,生长速率减缓[2]。因而维持肠上皮有序运转对提高营养物质利用效率和维持动物机体生长发育具有重要作用。
谷氨酸是肠上皮的主要能源物质[3],是与肠道黏膜生长和代谢相关的重要氨基酸之一[4-5]。作为一种功能性氨基酸,谷氨酸不仅参与蛋白质的合成和氧化供能[6],而且具有改善肠道完整性、增强肠道屏障功能和抗氧化能力、提高肠道干细胞的活性和促进肠道发育的作用[7-10]。
谷氨酸的高效利用有助于肠上皮更新或损伤后再生,因此,本文就肠道中谷氨酸转运和代谢、谷氨酸对肠上皮发育的影响及其可能的机制作一综述,以期为开发调控肠道发育的营养干预技术提供思路。
1 谷氨酸的吸收与转运研究表明,少量谷氨酸在胃内被吸收,其余大部分在肠道中被吸收[11]。小肠作为营养物质主要的吸收场所,其刷状缘膜上存在着各种营养物质的转运载体。进入小肠的谷氨酸经刷状缘膜上的转运载体进入肠上皮细胞内进行代谢,没有被细胞分解代谢的谷氨酸则通过基底膜上的转运载体进入血液循环供其他组织利用[12]。
谷氨酸转运系统包括XAG-和XC-转运系统(如图 1所示)。谷氨酸在转运过程中,首先通过高亲和力的Na+依赖性XAG-转运系统,将1个谷氨酸和3个Na+、1个H+结合,谷氨酸通过结合位点的转向运送到细胞内,同时伴有K+释放到细胞外;Na+被Na+-K+泵泵出到细胞外,以维持胞内外的离子平衡。XC-转运系统是通过将谷氨酸转运出细胞外置换胱氨酸进入细胞,是非Na+依赖性转运系统[13-14]。
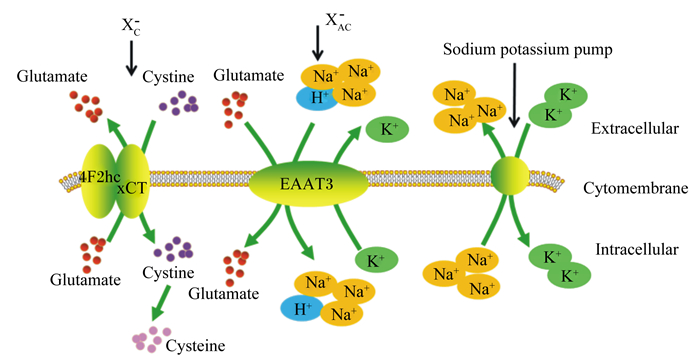
|
Glutamate:谷氨酸;Cysteine:半胱氨酸;Cystine:胱氨酸;Extracellular:细胞外;Cytomembrane:细胞膜;Intracellular:细胞内;Sodium potassium pump:钠钾泵; EAAT3: excitatory amino acid transporter 3。 图 1 谷氨酸转运系统 Fig. 1 Glutamate transport system[14] |
高亲和力的谷氨酸转运载体被分为3种主要类型:谷氨酸/天冬氨酸转运体(glutamate/aspartate transporter,GLAST)亦称兴奋性氨基酸转运子1(excitatory amino acid transporter 1,EAAT1)、神经胶质谷氨酸转运体1(glial glutamate transporter 1,GLT1)亦称兴奋性氨基酸转运子2(excitatory amino acid transporter 2,EAAT2)、兴奋性氨基酸载体1(excitatory amino acid carrier 1,EAAC1)亦称兴奋性氨基酸转运子3(excitatory amino acid transporter 3,EAAT3)[15]。通过这些转运载体及转运系统,谷氨酸可以完成从肠腔至肠上皮细胞再到血液循环的转运[14]。其中由溶质载体1(SLC1A1)编码的EAAT3是肠道中最重要的谷氨酸转运载体[15],肠腔中几乎所有的谷氨酸都是通过刷状缘膜上的EAAT3转运至肠上皮细胞内[16]。大量研究表明,小肠EAAT3基因表达与家鸡和家鸽胚胎期小肠生长水平密切相关[17-19]。胚胎期注射EAATs抑制剂L-反式-吡咯烷-2, 4-二羧酸(L-trans-pyrrolidine-2, 4-dicarboxylic acid,L-trans-PDC)可抑制鸡胚小肠发育[20]。在猪的饲粮中补充谷氨酸可提高谷氨酸转运载体在空肠的表达量,增加谷氨酸的转运效率,使机体更好地利用饲粮中的谷氨酸[21]。
2 谷氨酸在小肠中的代谢虽然大部分氨基酸在肝脏中进行代谢,但肠道也是谷氨酸、谷氨酰胺和天冬氨酸等氨基酸的重要代谢场所。谷氨酸可以作为中间代谢产物将氨基酸代谢和蛋白质代谢连接起来[22],在蛋白质合成和代谢方面起着重要作用[23]。肠腔中的细菌以及肠上皮细胞可利用谷氨酸生成谷胱甘肽等活性物质,还可以被转化成天冬氨酸、甘氨酸等其他非必需氨基酸[24-26],作为合成嘌呤或嘧啶核苷酸的前体物质。谷氨酸在小肠的代谢大多在肠上皮细胞中发生。进入肠上皮细胞内的谷氨酸的主要代谢方式是氧化,通过转氨作用生成α-酮戊二酸进入线粒体后,经三羧酸循环生成三磷酸腺苷(ATP),为肠上皮的有序运转提供能量[13]。谷氨酸代谢途径如图 2所示[27]。
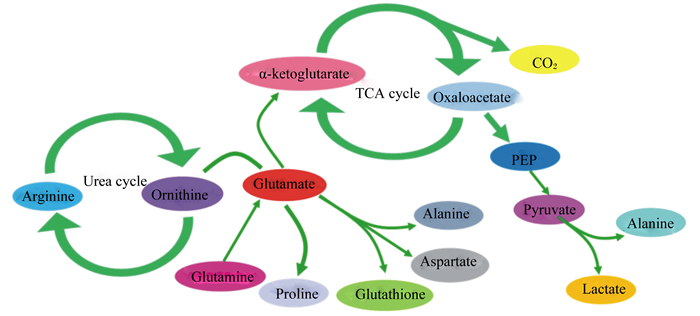
|
α-ketoglutarate:α-酮戊二酸;TCA cycle:三羧酸循环;Oxaloacetate:草酰乙酸;Arginine:精氨酸;Urea cycle:尿素循环;Ornithine:鸟氨酸;Glutamate:谷氨酸;PEP:磷酸烯醇式丙酮酸;Alanine:丙氨酸;Pyruvate:丙酮酸;Glutamine:谷氨酰胺;Proline:脯氨酸;Glutathione:谷胱甘肽;Aspartate:天冬氨酸;Lactate:乳酸。 图 2 谷氨酸的代谢途径 Fig. 2 Metabolic pathways of glutamate[27] |
值得注意的是,谷氨酰胺虽和谷氨酸一样具有为肠上皮细胞提供能量的功能,但是当谷氨酸和谷氨酰胺同时存在时,肠上皮细胞会优先利用谷氨酸,从而抑制谷氨酰胺氧化供能[25]。此外,有研究表明,内源性谷氨酰胺转化得到的谷氨酸不能用于合成谷胱甘肽,说明谷氨酰胺代谢产生的谷氨酸和从饲粮中获取的谷氨酸的代谢位置相互隔离,且不同来源的谷氨酸的代谢方式也不尽相同[28]。作为一种兴奋性神经递质,血液中高含量的谷氨酸会引起神经系统中毒。研究发现,经由肌肉注射的谷氨酸会引起大鼠血液中谷氨酸含量大幅上升,从而导致神经中毒。而饲喂谷氨酸含量是正常含量4倍的高谷氨酸饲粮,不会引起血液中谷氨酸含量的升高,提示经肠道吸收的谷氨酸大部分可能被代谢成其他非必需氨基酸或被氧化供能,进入血液中的谷氨酸含量有限,因此不会导致神经中毒[29]。
3 谷氨酸对哺乳动物雷帕霉素靶蛋白复合物1(mTORC1)信号通路的调控作用哺乳动物雷帕霉素靶蛋白(mTOR)是一种高度保守的非典型丝氨酸/苏氨酸蛋白激酶,可整合细胞外信号,磷酸化其下游靶蛋白核糖体蛋白S6激酶1(S6K1)和elF4E结合蛋白1(4EBP1),影响mRNA翻译,从而参与调控细胞生长、增殖等过程。其中mTORC1是细胞感知和整合外界信号刺激的关键,细胞蛋白质合成能力增强主要是由于mTORC1信号对氨基酸和胰岛素的敏感性增加[30]。目前亮氨酸[31-32]和精氨酸[33-34]进入细胞后,激活mTORC1的感应机制已经较为清楚,而谷氨酸被EAAT3转运至细胞内,如何将信号传导至mTORC1有待进一步解析。本实验室研究表明,谷氨酸可激活磷脂酰肌醇-3-羟激酶/蛋白激酶B/mTOR(PI3K/AKT/mTOR)通路。而用L-trans-PDC或无谷氨酸的培养基处理猪肠上皮细胞系IPEC-1,发现EAAT3、mTORC1信号通路关键蛋白质表达和细胞增殖均被抑制[35]。EAAT3作为谷氨酸的转运体,其是否同时是谷氨酸的感应体,介导谷氨酸调控激活mTORC1信号通路,一直未能得到证实。不过可以肯定的是,EAAT3表达量高低可影响谷氨酸的吸收,进入胞质中的谷氨酸可刺激mTORC1信号通路活性[36],进而影响蛋白质合成和细胞生长。
4 谷氨酸和mTORC1信号通路对肠道干细胞活性的影响肠道干细胞是驱动肠上皮内稳态平衡和再生的关键。包括2类肠道干细胞亚群,一类被称为+4干细胞,属于沉默型干细胞;另一类被称为CBC细胞,属于活跃型干细胞。B细胞特异性白血病病毒插入位点(Bmi1)和富含亮氨酸重复单位的G蛋白偶联受体5(Lgr5)分别是+4干细胞和CBC干细胞的标志基因[37-38]。每个小肠隐窝中仅含少量干细胞[39]。Metcalfe等[40]发现,经辐射诱导损伤的小鼠肠道,Lgr5+干细胞(具有Lgr5标记的干细胞)大部分丢失,但在随后的几天内通过激活Lgr5-干细胞(Lgr5+干细胞缺失可诱导处于静息状态的沉默型干细胞分化成不具有Lgr5标记的干细胞,即Lgr5-干细胞),隐窝绒毛轴得以再生。肠道干细胞活性受外在刺激和内在微环境信号的调控。细胞外刺激,包括营养素和环境因子等,均可引起干细胞快速应答以使机体适应外部环境的变化[41-42]。Deng等[8]通过在果蝇饲粮中分别添加谷氨酸、谷氨酰胺、天冬氨酸和葡萄糖等,发现仅谷氨酸可提高肠道干细胞的活性,并确认只有通过肠上皮摄取途径,才可以提高肠道干细胞活性和促进肠道发育。本实验室发现,谷氨酸可增加猪空肠隐窝干细胞扩增成为类肠团的生成效率和出芽指数。
肠道干细胞生活在由邻近的小肠细胞和隐窝周边间充质细胞等组成的微环境中。一般认为Wnt和Notch信号负责调控肠道干细胞活性和肠道稳态,是肠道上皮细胞有序结构的控制器[43-45]。Xiong等[46]对仔猪肠上皮隐窝绒毛轴进行分级、分离检测发现,mTORC1信号与Wnt和Notch信号相似,富集于隐窝微环境,并沿这小肠隐窝-绒毛轴逐渐递减。此外,有研究发现,mTORC1信号同样参与了肠道干细胞和祖细胞的代谢,并增加其活性,加速肠上皮损伤修复过程。当mTORC1缺失时,小鼠肠道干细胞扩增能力下降,肠上皮更新进程发生紊乱,绒毛变短,损伤后自修复终止[47]。
谷氨酸增强肠道干细胞活性是否是通过激活mTORC1信号通路实现,mTORC1信号又是如何在肠道干细胞微环境中发挥作用缺乏进一步的研究。对这些问题的解析有助于提高谷氨酸和mTORC1靶向治疗的有效性和特异性,完善肠道干细胞调控和肠上皮发育的机制。
5 谷氨酸对肠黏膜屏障功能的影响肠道是机体与外界沟通的媒介,同样也是外界有毒有害物质攻击的首要靶标之一。毒素、环境因子和断奶应激都会引起动物肠道结构和功能损伤,危害肠道健康,降低动物生产性能[48-51]。而谷氨酸可改善肠道完整性,缓解肠道损伤[9-10]。谷氨酸对肠上皮的保护作用体现在维持肠道屏障功能正常运转,主要包括肠黏膜机械屏障、化学屏障和免疫屏障。
5.1 机械屏障肠道黏膜机械屏障功能主要由完整的肠上皮细胞及细胞间的紧密连接组成。其中肠上皮细胞可选择性转运某些物质进入内环境中,细胞间紧密连接则可连接相邻2个细胞骨架,封闭细胞间的间隙,在维持机械屏障中发挥非常重要的作用[9, 52]。研究发现,谷氨酸可促进肠上皮细胞增殖[53]。饲粮中添加1%谷氨酸,可增加断奶仔猪空肠绒毛高度和肠道黏膜厚度,改善肠道完整性,增强机械屏障[8]。此外,谷氨酸能增加肠上皮细胞跨膜电阻值,降低其通透性,提高紧密连接蛋白[闭合蛋白(occludin)、封闭蛋白(claudin)、胞浆蛋白(ZOs)]表达[7, 54]。
5.2 化学屏障肠道化学屏障主要由覆盖在肠上皮细胞上的疏松黏液层和黏液中的黏蛋白组成。肠道黏液层是抵御外界病原体的第1道防线,可抑制病原微生物与上皮细胞直接作用,而黏蛋白是肠道黏液层的主要组成成分[55]。谷氨酸可通过肠道谷氨酸受体,包括钙敏感受体(CaSRs)、代谢型谷氨酸受体(mGluRs),刺激一氧化氮(NO)的合成,促进五羟色胺(5-HT)释放,激活迷走神经,将肠道内的信息传递至中枢神经系统,中枢神经系统接受并整合信息后调节肠道中黏液、HCO3-的分泌[56-60]。
黏蛋白具有防止致病菌黏附和侵袭肠道的作用[61]。Amagase等[62]研究发现,谷氨酸可逆转氯索洛芬引起的大鼠小肠黏蛋白2(MUC2)基因及蛋白质表达降低,增加黏液层厚度,加强肠道化学屏障功能。黏液-碳酸氢盐屏障也是肠黏膜化学屏障的组成部分。大量研究表明,大鼠胃肠道内灌注不同剂量的谷氨酸(0.1~10.0 mmol/L)可增加十二指肠HCO3-的分泌,使十二指肠的pH及黏液层的厚度出现剂量依赖性增加,从而减少了酸性环境以及细菌对肠上皮细胞的侵害,保护黏液-碳酸氢盐屏障功能[63-66]。有趣的是,在氨基酸丰富的饲粮中添加谷氨酸会增强犬胃液和胃蛋白酶的分泌,而当其添加于碳水化合物丰富的饲粮中时则不会影响犬胃液的分泌[22-23]。这表明在动物摄入大量蛋白质类食物时,谷氨酸可以通过调控胃液的分泌调控肠道pH,维持肠黏膜化学屏障功能正常。
5.3 免疫屏障肠黏膜免疫屏障是由肠道相关淋巴样组织(GALT)、肠系膜淋巴结(ETC)、分泌型抗体构成的可以对抗毒素、抗原及潜在的有害生物侵害的重要屏障[62]。其中肠道相关淋巴样组织在保护机体免受外来抗原和病原体侵害的同时,允许肠道免疫系统对肠道共生菌有一定的耐受性[67]。
研究表明,T细胞、B细胞、树枝状细胞及巨噬细胞均表达谷氨酸受体[68-69],谷氨酸可使体外培养的B细胞及外周单核细胞产生更多的免疫球蛋白(Ig)G与IgE[69]。Pacheco等[70]和Ruth等[71]发现在抗原接触T细胞时,谷氨酸会由树枝状细胞释放,从而影响T细胞的增殖与细胞因子的产生,根据以上研究结果推测,谷氨酸可能通过调节肠道B细胞和T细胞功能增强肠道的免疫机能。
6 谷氨酸对肠道抗氧化能力的影响以谷氨酸为底物合成的谷胱甘肽具有较强的抗氧化作用,可降低氧化应激诱导的肠道损伤。研究发现,饲粮中添加2%谷氨酸可缓解过氧化氢导致的仔猪肠道氨基酸吸收和氧化还原反应紊乱[72]。刘明锋[73]研究同样发现,饲粮中添加1%的谷氨酸能够有效增加肠道的抗氧化能力,缓解敌草快这一氧化应激源引起的血清中苏氨酸和蛋氨酸含量下降。铜离子能引起草鱼肠上皮细胞活性氧的产生,而谷氨酸预处理后可有效防止其被氧化损伤,降低总超氧化物歧化酶(T-SOD)、谷胱甘肽转移酶(GST)、谷胱甘肽还原酶(GR)和抗超氧化物阴离子(ASA)在内的一系列抗氧化物的消耗[74]。
7 小结综上所述,谷氨酸通过激活mTORC1信号通路调控肠上皮细胞生长,提高干细胞活性,增强肠道抗氧化能力,维持肠上皮完整性,完善肠道屏障功能。但是人们尚未明晰谷氨酸如何作用于细胞膜上的感应体启动细胞内信号,并将信号传导至mTORC1以调控蛋白质合成等过程。同时,对于谷氨酸如何调节肠道干细胞活性,且这一过程与mTORC1信号通路之间的关联性也有待进一步探究。由于谷氨酸在肠道能量供应和营养调控中发挥的重要作用,谷氨酸功能挖掘越来越受到关注。因此,有必要深入解析谷氨酸调节肠上皮发育的信号网络和促进肠上皮发育的机制,为其在畜牧生产中调控动物肠道发育的生产应用和在营养学或药理学方面对疾病进行干预和治疗提供理论依据。
| [1] |
HANKE D, POHLMANN A, SAUTER-LOUIS C, et al. Porcine epidemic diarrhea in europe:in-detail analyses of disease dynamics and molecular epidemiology[J]. Viruses, 2017, 9(7): 177. DOI:10.3390/v9070177 |
| [2] |
TULEMISOVA Z, BIYASHEV K, BIYASHEV B, et al. Prophilaxy of gasstro-intestinal diseases of young animals[J]. Journal of Animal and Veterinary Advances, 2013, 12(22): 1645-1650. |
| [3] |
BROSNAN J T, BROSNAN M E. Glutamate:a truly functional amino acid[J]. Amino Acids, 2013, 45(3): 413-418. DOI:10.1007/s00726-012-1280-4 |
| [4] |
FAN M Z, MATTHEWS J C, ETIENNE N M, et al. Expression of apical membrane L-glutamate transporters in neonatal porcine epithelial cells along the small intestinal crypt-villus axis[J]. American Journal of Physiology:Gastrointestinal and Liver Physiology, 2004, 287(2): G385-G398. DOI:10.1152/ajpgi.00232.2003 |
| [5] |
LIN M, ZHANG B L, YU C N, et al. L-glutamate supplementation improves small intestinal architecture and enhances the expressions of jejunal mucosa amino acid receptors and transporters in weaning piglets[J]. PLoS One, 2014, 9(11): e111950. DOI:10.1371/journal.pone.0111950 |
| [6] |
YIN J, LIU M F, REN W K, et al. Effects of dietary supplementation with glutamate and aspartate on diquat-induced oxidative stress in piglets[J]. PLoS One, 2015, 10(4): e0122893. DOI:10.1371/journal.pone.0122893 |
| [7] |
JIAO N, WU Z L, JI Y, et al. L-glutamate enhances barrier and antioxidative functions in intestinal porcine epithelial cells[J]. The Journal of Nutrition, 2015, 145(10): 2258-2264. DOI:10.3945/jn.115.217661 |
| [8] |
DENG H S, GERENCSER A A, JASPER H. Signal integration by Ca2+ regulates intestinal stem-cell activity[J]. Nature, 2015, 528(7581): 212-217. DOI:10.1038/nature16170 |
| [9] |
TURNER J R. Intestinal mucosal barrier function in health and disease[J]. Nature Reviews Immunology, 2009, 9(11): 799-809. DOI:10.1038/nri2653 |
| [10] |
WU X, ZHANG Y, LIU Z, et al. Effects of oral supplementation with glutamate or combination of glutamate and N-carbamylglutamate on intestinal mucosa morphology and epithelium cell proliferation in weanling piglets[J]. Journal of Animal Science, 2012, 90(Suppl.4): 337-339. |
| [11] |
TORⅡ K. Physiological roles of dietary glutamate signaling via gut-brain axis for energy expenditure[J]. Journal of Gastroenterology, 2013, 48(4): 442-451. DOI:10.1007/s00535-013-0778-1 |
| [12] |
姜廷波.葡萄糖和赖氨酸水平对仔猪小肠上皮细胞赖氨酸吸收、转运的影响[D].硕士学位论文.长春: 吉林农业大学, 2017. http://cdmd.cnki.com.cn/Article/CDMD-10193-1017842933.htm
|
| [13] |
张军.谷氨酸钠在仔猪胃肠道的转运和代谢及机制研究[D].博士学位论文.北京: 中国科学院大学, 2013. http://www.wanfangdata.com.cn/details/detail.do?_type=degree&id=Y2368964
|
| [14] |
周济宏, 李幼生, 黎介寿. 谷氨酰胺转运载体的种类与分布及功能特点[J]. 医学研究生学报, 2007(04): 408-411. DOI:10.3969/j.issn.1008-8199.2007.04.021 |
| [15] |
HU Q X, OTTESTAD-HANSEN S, HOLMSETH S, et al. Expression of glutamate transporters in mouse liver, kidney, and intestine[J]. Journal of Histochemistry & Cytochemistry, 2018, 66(3): 189-202. |
| [16] |
FAN M Z, MATTHEWS J C, ETIENNE N M P, et al. Expression of apical membrane L-glutamate transporters in neonatal porcine epithelial cells along the small intestinal crypt-villus axis[J]. American Journal of Physiology:Gastrointestinal and Liver Physiology, 2004, 287(2): G385-G398. DOI:10.1152/ajpgi.00232.2003 |
| [17] |
CHEN M X, LI X G, YAN H C, et al. Effect of egg weight on composition, embryonic growth, and expression of amino acid transporter genes in yolk sac membranes and small intestines of the domestic pigeon (Columba livia)[J]. Poultry Science, 2016, 95(6): 1425-1432. DOI:10.3382/ps/pew044 |
| [18] |
CHEN M X, LI X G, YANG J X, et al. Growth of embryo and gene expression of nutrient transporters in the small intestine of the domestic pigeon (Columba livia)[J]. Journal of Zhejiang University (SCIENCE B), 2015, 16(6): 511-523. DOI:10.1631/jzus.B1400340 |
| [19] |
LI X G, CHEN X L, WANG X Q. Changes in relative organ weights and intestinal transporter gene expression in embryos from white olymouth rock and wens yellow feather chickens[J]. Comparative Biochemistry and Physiology Part A:Molecular & Integrative Physiology, 2013, 164(2): 368-375. |
| [20] |
LI X G, SUI W G, YAN H C, et al. The in ovo administration of L-trans pyrrolidine-2, 4-dicarboxylic acid regulates small intestinal growth in chicks[J]. Animal, 2014, 8(10): 1677-1683. DOI:10.1017/S1751731114001645 |
| [21] |
ZHANG J, YIN Y L, SHU X G, et al. Oral administration of MSG increases expression of glutamate receptors and transporters in the gastrointestinal tract of young piglets[J]. Amino Acids, 2013, 45(5): 1169-1177. DOI:10.1007/s00726-013-1573-2 |
| [22] |
BERGEN W G, WU G Y. Intestinal nitrogen recycling and utilization in health and disease[J]. The Journal of Nutrition, 2009, 139(5): 821-825. DOI:10.3945/jn.109.104497 |
| [23] |
WU G Y, BAZER F W, DAVIS T A, et al. Arginine metabolism and nutrition in growth, health and disease[J]. Amino Acids, 2009, 37(1): 153-168. DOI:10.1007/s00726-008-0210-y |
| [24] |
KANAI Y, HEDIGER M A. The glutamate and neutral amino acid transporter family:physiological and pharmacological implications[J]. European Journal of Pharmacology, 2003, 479(1/2/3): 237-247. |
| [25] |
BLACHIER F, MARIOTTI F, HUNEAU J F, et al. Effects of amino acid-derived luminal metabolites on the colonic epithelium and physiopathological consequences[J]. Amino Acids, 2007, 33(4): 547-562. DOI:10.1007/s00726-006-0477-9 |
| [26] |
CREMIN J D, Jr., FITCH M D, FLEMING S. Glucose alleviates ammonia-induced inhibition of short-chain fatty acid metabolism in rat colonic epithelial cells[J]. American Journal of Physiology:Gastrointestinal and Liver Physiology, 2003, 25(1): G105-G114. |
| [27] |
BURRIN D G, STOLL B. Metabolic fate and function of dietary glutamate in the gut[J]. The American Journal of Clinical Nutrition, 2009, 90(3): 850S-860S. DOI:10.3945/ajcn.2009.27462Y |
| [28] |
REEDS P J, BURRIN D G, STOLL B, et al. Enteral glutamate is the preferential source for mucosal glutathione synthesis in fed piglets[J]. American Journal of Physiology, 1997, 273(2): E408-E415. |
| [29] |
BURRIN D G, STOLL B, FERNSTROM J D, et al. Metabolic fate and function of dietary glutamate in the gut[J]. American Journal of Clinical Nutrition, 2009, 90(3): 850S-856S. DOI:10.3945/ajcn.2009.27462Y |
| [30] |
SURYAWAN A, ORELLANA R A, NGUYEN H V, et al. Activation by insulin and amino acids of signaling components leading to translation initiation in skeletal muscle of neonatal pigs is developmentally regulated[J]. American Journal of Physiology:Endocrinology and Metabolism, 2007, 293(6): E1597-E1605. DOI:10.1152/ajpendo.00307.2007 |
| [31] |
WOLFSON R L, CHANTRANUPONG L, SAXTON R A, et al. Sestrin 2 is a leucine sensor for the mTORC1 pathway[J]. Science, 2016, 351(6268): 43-48. DOI:10.1126/science.aab2674 |
| [32] |
HAN J M, JEONG S J, PARK M C, et al. Leucyl-tRNA synthetase is an intracellular leucine sensor for the mTORC1-signaling pathway[J]. Cell, 2012, 149(2): 410-424. DOI:10.1016/j.cell.2012.02.044 |
| [33] |
CHANTRANUPONG L, SCARIA S M, SAXTON R A, et al. The castor proteins are arginine sensors for the mtorc1 pathway[J]. Cell, 2016, 165(1): 153-164. DOI:10.1016/j.cell.2016.02.035 |
| [34] |
WANG S Y, TSUN Z Y, WOLFSON R L, et al. Metabolism.Lysosomal amino acid transporter SLC38A9 signals arginine sufficiency to mTORC1[J]. Science, 2015, 347(6218): 188-194. DOI:10.1126/science.1257132 |
| [35] |
LI X G, SUI W G, GAO C Q, et al. L-glutamate deficiency can trigger proliferation inhibition via down regulation of the mTOR/S6K1 pathway in pig intestinal epithelial cells[J]. Journal of Animal Science, 2016, 94(4): 1541-1549. DOI:10.2527/jas.2015-9432 |
| [36] |
YE J L, GAO C Q, LI X G, et al. EAAT3 promotes amino acid transport and proliferation of porcine intestinal epithelial cells[J]. Oncotarget, 2016, 7(25): 38681-38692. |
| [37] |
CLEVERS H. Stem cells:a unifying theory for the crypt[J]. Nature, 2013, 495(7439): 53-54. DOI:10.1038/nature11958 |
| [38] |
YAN K S, CHIA L A, LI X N, et al. The intestinal stem cell markers Bmi1 and Lgr5 identify two functionally distinct populations[J]. Proceedings of the National Academy of Sciences of the United States of America, 2012, 109(2): 466-471. DOI:10.1073/pnas.1118857109 |
| [39] |
UMAR S. Intestinal stem cells[J]. Current Gastroenterology Reports, 2010, 12(5): 340-348. DOI:10.1007/s11894-010-0130-3 |
| [40] |
METCALFE C, KLJAVIN N M, YBARRA R, et al. Lgr5+ stem cells are indispensable for radiation-induced intestinal regeneration[J]. Cell Stem Cell, 2014, 14(2): 149-159. DOI:10.1016/j.stem.2013.11.008 |
| [41] |
O'BRIEN L E, SOLIMAN S S, LI X H, et al. Altered modes of stem cell division drive adaptive intestinal growth[J]. Cell, 2011, 147(3): 603-614. DOI:10.1016/j.cell.2011.08.048 |
| [42] |
DAILEY M J. Nutrient-induced intestinal adaption and its effect in obesity[J]. Physiology & Behavior, 2014, 136: 74-78. |
| [43] |
KRETZSCHMAR K, CLEVERS H. Wnt/β-catenin signaling in adult mammalian epithelial stem cells[J]. Developmental Biology, 2017, 428(2): 273-282. DOI:10.1016/j.ydbio.2017.05.015 |
| [44] |
ZOU W Y, BLUTT S E, ZENG X L, et al. Epithelial WNT ligands are essential drivers of intestinal stem cell activation[J]. Cell Reports, 2018, 22(4): 1003-1015. DOI:10.1016/j.celrep.2017.12.093 |
| [45] |
YIN X L, FARIN H F, ES J H V, et al. Niche-independent high-purity cultures of Lgr5+ intestinal stem cells and their progeny[J]. Nature Methods, 2014, 11(1): 106-112. DOI:10.1038/nmeth.2737 |
| [46] |
XIONG X, YANG H S, TAN B, et al. Differential expression of proteins involved in energy production along the crypt-villus axis in early-weaning pig small intestine[J]. American Journal of Physiology:Gastrointestinal and Liver Physiology, 2015, 309(4): G229-G237. DOI:10.1152/ajpgi.00095.2015 |
| [47] |
SAMPSON L L, DAVIS A K, GROGG M W, et al. mTOR disruption causes intestinal epithelial cell defects and intestinal atrophy postinjury in mice[J]. The FASEB Journal, 2015, 30(3): 1263-1275. |
| [48] |
MILLER B G, NEWBY T J, STOKES C R, et al. The importance of dietary antigen in the cause of postweaning diarrhea in pigs[J]. American Journal of Veterinary Research, 1984, 45(9): 1730-1733. |
| [49] |
HAMPSON D J. Alterations in piglet small intestinal structure at weaning[J]. Research in Veterinary Science, 1986, 40(1): 32-40. DOI:10.1016/S0034-5288(18)30482-X |
| [50] |
VAN BEERS-SCHREURS H M G, NABUURS M J A, VELLENGA L, et al. Weaning and the weanling diet influence the villous height and crypt depth in the small intestine of pigs and alter the concentrations of short-chain fatty acids in the large intestine and blood[J]. The Journal of Nutrition, 1998, 128(6): 947-953. DOI:10.1093/jn/128.6.947 |
| [51] |
TONG M B, LAARVELD A G, VAN KESSEL D L, et al. Effect of segregated early weaning on postweaning small intestinal development in pigs[J]. Journal of Animal Science, 1999, 77(12): 3191-3200. DOI:10.2527/1999.77123191x |
| [52] |
CAMILLERI M, MADSEN K, SPILLER R, et al. Intestinal barrier function in health and gastrointestinal disease[J]. Neurogastroenterology & Motility, 2012, 24(6): 503-512. |
| [53] |
XIAO W D, FENG Y J, HOLST J J, et al. Glutamate prevents intestinal atrophy via luminal nutrient sensing in a mouse model of total parenteral nutrition[J]. The FASEB Journal, 2014, 28(5): 2073-2087. DOI:10.1096/fj.13-238311 |
| [54] |
VERMEULEN M A, DE JONG J, VAESSEN M J, et al. Glutamate reduces experimental intestinal hyperpermeability and facilitates glutamine support of gut integrity[J]. World Journal of Gastroenterology, 2011, 17(12): 1569-1573. DOI:10.3748/wjg.v17.i12.1569 |
| [55] |
MERGA Y, CAMPBELL B J, RHODES J M. Mucosal barrier, bacteria and inflammatory bowel disease:possibilities for therapy[J]. Digestive Diseases, 2014, 32(4): 475-483. DOI:10.1159/000358156 |
| [56] |
DU J, LI X H, LI Y J. Glutamate in peripheral organs:biology and pharmacology[J]. European Journal of Pharmacology, 2016, 784: 42-48. DOI:10.1016/j.ejphar.2016.05.009 |
| [57] |
BEZENÇON C, LE COUTRE J, DAMAK S. Taste-signaling proteins are coexpressed in solitary intestinal epithelial cells[J]. Chemical Senses, 2007, 32(1): 41-49. DOI:10.1093/chemse/bjl034 |
| [58] |
NAKAMURA E, HASUMURA M, SAN GABRIEL A, et al. New frontiers in gut nutrient sensor research:luminal glutamate-sensing cells in rat gastric mucosa[J]. Journal of Pharmacological Sciences, 2010, 112(1): 13-18. DOI:10.1254/jphs.09R16FM |
| [59] |
TORⅡ K, UNEYAMA H, NAKAMURA E. Physiological roles of dietary glutamate signaling via gut-brain axis due to efficient digestion and absorption[J]. Journal of Gastroenterology, 2013, 48(4): 442-451. DOI:10.1007/s00535-013-0778-1 |
| [60] |
KITAMURA A, TSURUGIZAWA T, UEMATSU A, et al. New therapeutic strategy for amino acid medicine:effects of dietary glutamate on gut and brain function[J]. Journal of Pharmacological Sciences, 2012, 118(2): 138-144. DOI:10.1254/jphs.11R06FM |
| [61] |
JOHANSSON M E V, PHILLIPSON M, PETERSSON J, et al. The inner of the two Muc2 mucin-dependent mucus layers in colon is devoid of bacteria[J]. Proceedings of the National Academy of Sciences of the United States of America, 2008, 105(39): 15064-15069. DOI:10.1073/pnas.0803124105 |
| [62] |
AMAGASE K, KIMURA Y, WADA A, et al. Prophylactic effect of monosodium glutamate on NSAID-induced enteropathy in rats[J]. Current Pharmaceutical Design, 2014, 20(16): 2783-2790. DOI:10.2174/13816128113199990579 |
| [63] |
AKIBA Y, WATANABE C, MIZUMORI M, et al. Luminal L-glutamate enhances duodenal mucosal defense mechanisms via multiple glutamate receptors in rats[J]. American Journal of Physiology:Gastrointestinal and Liver Physiology, 2009, 297(4): G781-G791. DOI:10.1152/ajpgi.90605.2008 |
| [64] |
AKIBA Y, KAUNITZ J D. Duodenal chemosensing and mucosal defenses[J]. Digestion, 2011, 83(Suppl.1): 25-31. |
| [65] |
UNEYAMA H, NIIJIMA A, SAN GABRIEL A, et al. Luminal amino acid sensing in the rat gastric mucosa[J]. American Journal of Physiology:Gastrointestinal and Liver Physiology, 2006, 291(6): G1163-G1170. DOI:10.1152/ajpgi.00587.2005 |
| [66] |
AKIBA Y, KAUNITZ J D. Luminal chemosensing and upper gastrointestinal mucosal defenses[J]. The American Journal of Clinical Nutrition, 2009, 90(3): 826S-831S. DOI:10.3945/ajcn.2009.27462U |
| [67] |
左增妍, 张彩. 肠道黏膜免疫耐受机制研究进展[J]. 现代免疫学, 2015, 35(1): 68-71. |
| [68] |
REZZANI R, CORSETTI G, RODELLA L, et al. Cyclosporine-A treatment inhibits the expression of metabotropic glutamate receptors in rat thymus[J]. Acta Histochemica, 2003, 105(1): 81-87. DOI:10.1078/0065-1281-00688 |
| [69] |
STURGILL J L, MATHEWS J, SCHERLE P, et al. Glutamate signaling through the kainate receptor enhances human immunoglobulin production[J]. Journal of Neuroimmunology, 2011, 233(1/2): 80-89. |
| [70] |
PACHECO R, OLIVA H, MARTINEZ-NAVÍO J M, et al. Glutamate released by dendritic cells as a novel modulator of T cell activation[J]. The Journal of Immunology, 2006, 177(10): 6695-6704. DOI:10.4049/jimmunol.177.10.6695 |
| [71] |
RUTH M R, FIELD C J. The immune modifying effects of amino acids on gut-associated lymphoid tissue[J]. Journal of Animal Science and Biotechnology, 2013, 4(1): 27. DOI:10.1186/2049-1891-4-27 |
| [72] |
段杰林.酸性氨基酸缓解过氧化氢介导仔猪肠道氧化损伤机制研究[D].硕士学位论文.北京: 中国科学院大学, 2016. http://www.wanfangdata.com.cn/details/detail.do?_type=degree&id=Y3056195
|
| [73] |
刘明锋.谷氨酸对断奶仔猪抗氧化能力的影响研究[D].硕士学位论文.长沙: 湖南农业大学, 2014. http://cdmd.cnki.com.cn/Article/CDMD-10537-1015602385.htm
|
| [74] |
JIANG J, WU X Y, ZHOU X Q, et al. Glutamate ameliorates copper-induced oxidative injury by regulating antioxidant defences in fish intestine[J]. British Journal of Nutrition, 2016, 116(1): 70-79. DOI:10.1017/S0007114516001732 |




